Answerįractionation can also occur from fractional crystallization. What will be the composition of the two resulting fractions? Answers.Īccept or Reject: the longer we let the melting go on before separating the fractions, the more Na rich the one fraction becomes and the more Ca rich the other fraction becomes. Imagine the system heats up to 1350 o at which point it fractionates. In each case, one fraction would be more Na rich than what we began with, and the second fraction would be more Ca rich. We could, of course, separate the melt from the unmelted crystal at any time during the process before the last bit of crystal melts, and fractionated the system. We have divided the original composition (50% anorthite) into two fractions, a Na rich melt (about 9% anorthite) and a slightly Na impoverished crystal (about 49% anorthite.) Now imagine that this first melt is removed from the system so it can no longer react. If the system remained in equilibrium at all times, what will be the composition of the crystals just before the last drop of melt crystallizes? Answer.
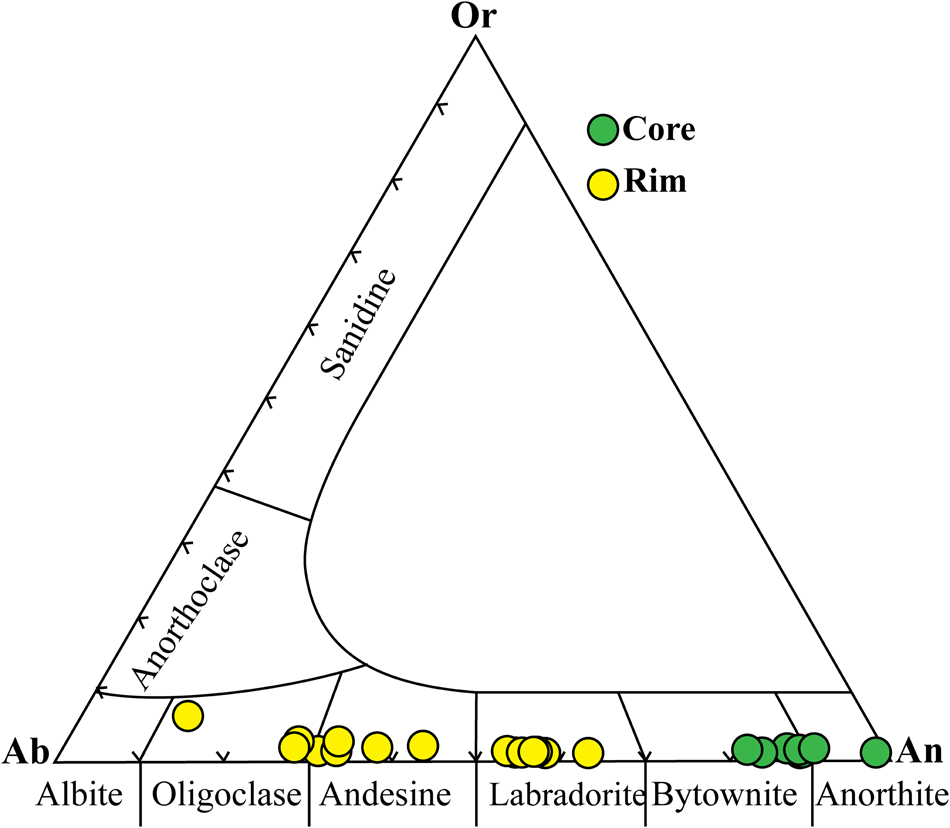
What will be the composition of the last drop of melt? And at what temperature? Answers.

The composition of the melt and crystals move down in tandem, exactly opposite each other along a horizontal line. The earlier formed crystals react with the melt, exchanging Ca for Na, to come to a composition in equilibrium with the temperature at the moment. But at the same time the composition of the crystals forming are moving down the solidus line (that is, decreasing in Ca.) This is because the system cools slowly enough to stay in equilibrium at all times. Thus, as the temperature lowers the composition of the melt migrates down the liquidus line. The system remains in equilibrium throughout its history so that all reactions can take place and everything can come to stability.Įverything in the original melt remains in communication throughout the crystallization process.īut observe, as crystallization continues Ca is removed from the melt faster than Na (a 30% melt yields a first crystal not at 30% but at 72%.) As a result as crystallization proceeds Na concentration gets higher in the melt and Ca gets lower.ĥ. Pressure is held constant at 1 atmosphere.Ĭomplete miscibility (mixability) in both liquid (magma) and crystal phases. One variable - Temperature plotted along the vertical axis.
The larger Ca (99A) is higher/hotter than the smaller Na (95A). NOTE that anorthite crystallizes at a much higher temperature than albite, just what we expect from Bowen's reaction series. Two components: high temperature CaAl 2Si 2O 8 (anorthite) and low temperature NaAl 2Si 2O 8 (albite) plotted along the horizontal axis. The conventions for the phase diagram include the following (illustration below): We want to observe the behavior of plagioclase under two conditions, one of complete equilibrium during crystallization, and the second of disequilibrium when fractionation occurs and igneous rocks evolve. The example here is for the plagioclase feldspars. The solid solution phase diagram explains the behavior of chemical solid solution series, such as the transition from high temperature, calcium-rich plagioclase to low temperature sodium-rich plagioclase, or the transition from high temperature magnesium-rich to low temperature iron-rich crystals in ferromagnesium minerals (e.g.
